Formaldehyde (HCHO) is a common substance in both indoor and outdoor environments. It is used in a wide range of products including wall board, glues, some carpets, fragrance products and insulation. It is also found in cigarette and cigar smoke and is a byproduct of hydrocarbon combustion. The National Toxicology Program (NTP) carcinogenic classification is that it is: “reasonably anticipated to be a human carcinogen.” Other health effects include eye, nose and skin irritation; exacerbation of asthma symptoms (characterized by coughing, difficulty breathing, bronchial spasm and pulmonary edema); lung damage; membranous nephropathy and liver toxicity. (Source: U.S. Department of Labor — www.osha.gov).
Formaldehyde exposure limits have been set by various governmental bodies. The National Institute for Occupational Safety and Health (NIOSH) has set a ceiling of 100 ppb for a 15 minute exposure.
Some manufacturers of ozone generators and ionizers that produce ozone as a byproduct claim that their devices decrease gaseous pollutants including formaldehyde. Research studies have shown that ozone can actually increase formaldehyde levels. In the early 1990,s Charles Weschler (Weschler et al., 1992) found that formaldehydes and other aldehydes increased with ozone exposure in a newly carpeted test chamber. In fact, even though some of the original VOC’s coming from the carpeting were eliminated the total VOC’s in the chamber increased. Mark Mason of the EPA National Management Research Laboratory (Mason, 2004) found that formaldehyde was produced when plug-in air fresheners were used in the presence of ozone. Morrison and Nazaroff (Morrison and Nazaroff, 2002a) also tested the effects of ozone on new and aired carpeting. They found increases in formaldehyde and other aldehydes.
Recently, The Sharper Image Corporation, marketers of the Ionic Breeze air cleaners, commissioned an independent project conducted by Columbia Analytical Services of Simi Valley, California. The project was entitled “Evaluation of the Sharper Image Quadra Air Cleaner Environmental Tobacco Smoke Trial.” The project draft was presented by Sharper Image Corporation as evidence of the “effectiveness” of their product in their suit against Consumers Union. Sharper Image had sued the publisher of the magazine, Consumers Reports, over their articles that found the Ionic Breeze “to be quiet but ineffective.” In November of 2004 U. S. District Judge Maxine Chesney dismissed the lawsuit stating that “Sharper Image had not demonstrated a reasonable probability that any of the challenged statements were false.” Sharper Image was ordered to pay attorneys fees for Consumers Union which were estimated to be over $400,000.
The Ionic Breeze is an ionizer that produces ozone as a byproduct. The Evaluation of Columbia Analytical Services was conducted by “smoking” cigarettes in two chambers — one with an Ionic Breeze and the other was used as a control. Measurements were taken over an eight hour period of: particle counts, carbon monoxide, Nicotene, solanesol, acrolein, benzene, styrene, formaldehyde and ozone. An odor evaluation was also conducted by an “odor panel” of lab personnel.
Particle counts showed some reduction of particles in the 1 and 2 micron sizes with the Ionic Breeze. A substantial INCREASE in particle counts were recorded in the 0.3 micron range. Other studies (Weschler, Sarwar) have found the same phenomenon when ozone reacts with VOC’s. This “generation” of sub-micron sized particles happens through chemical reactions of smaller particles and compounds that coagulate or coalesce into larger “measurable” ranges. These particles are a potential health hazard in that they are too small to be stopped by humans’ respiratory filters. Studies have shown that increases in outdoor sub-micron sized particles are directly related to increased mortality (Six cities study).
Several chemical compounds that are specific to Environmental Tobacco Smoke (ETS) were also evaluated. These markers included nicotine and 3-ethenylpyridine, which are specific to the vapor phase of ETS and solanesol, a marker for the particulate phase. Airborne concentrations of nicotine and 3-ethenylpyridine decreased at similar rates with and without the device operating. Solanesol decreased at a faster rate in the chamber with the Ionic Breeze. Carbon monoxide, acrolein, benzene and styrene all decreased at a similar rate with and without the device operating.
However, formaldehyde increased substantially (over 50%) in the chamber where the Ionic Breeze was operating. In the control chamber the airborne concentration of formaldehyde was essentially unchanged (64 parts per billion to 66 parts per billion). Formaldehyde in the chamber with the Ionic Breeze went from 82 parts per billion to 123 parts per billion at the end of the 8 hour test. Much of the increase (25 parts per billion) occurred in the last 4 hours of the test. Given this increasing trend, the formaldehyde level could have easily gone higher if the test had been continued over a longer period.
Studies have shown that ozone is consumed through chemical reactions with Volatile Organic Compounds (VOC’s). (Weschler, Corsi) The ozone levels in both chambers remained below 10 parts per billion throughout the test. The Ionic Breeze produces ozone at a rate of 2 to 3 milligrams per hour. In a chamber of this size one would anticipate an increase in ozone levels of at least 20 parts per billion (ie. From 10 parts per billion to 30 parts per billion). The fact that the ozone level did not increase is an indication that the ozone produced by the Ionic Breeze was consumed by the chemical reactions with the various components of the Environmental Tobacco Smoke.
The FDA has set limits for ozone for medical devices that produce ozone. The limits are 50 parts per billion coming from the machine or not more than 50 parts per billion accumulation in a given space. The reactions of ozone with other gaseous components of indoor air make the later limit relatively meaningless. The accumulation of ozone will vary considerably based on the chemical reactions where the ozone is consumed. The products of these reactions (such as formaldehyde) could easily be more dangerous than the ozone itself.
The test also measured the odor levels in both chambers. The following graph gives the results.
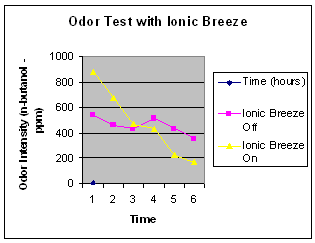
The initial intensity of the odor was greater in the chamber with the Ionic Breeze running. This finding is consistent with other research. Morrison and Nazaroff have shown that ozone reacts with VOC’s to create aldehydes and ketones with very low odor thresholds. It is very possible that such reactions could be the cause of this strong odor perception. More analysis of the components of the gasses created would have to be conducted to confirm this supposition. After approximately one hour the intensity of the odor in the chamber with the Ionic Breeze on was less than the control. After 8 hours the odor in the chamber with the Ionic Breeze on was 50% less than the control.
The fact that the odor decreased over time is interesting in that it was found that many of the chemicals in the ETS did not decrease proportionately. They generally either remained the same or increased over time. Odor reduction does not correlate well with chemical reduction. In other words, the perception of the elimination of the components of a substance (ETS) because of the decrease in odor of this substance (ETS) is not borne out by the results of these experiments. It seems that the human sense of smell is not a very good indicator of the effectiveness of an “air cleaner.”
Ozone generators and ionizers that produce ozone as a byproduct are not the only sources of indoor ozone. Ozone is produced by photo copiers, laser printers, and other office machines. Electronic precipitators, UV lights and other products that produce ozone are being installed in HVAC systems. Outdoor to indoor ozone transfers also occur. Indoor ozone levels usually measure between 30% and 70% of outdoor concentrations. As illustrated by these experiments even relatively low levels of ozone added to an indoor environment, change the chemistry of that indoor environment. The resulting chemical reactions produce sub-micron sized particles (secondary aerosols). The chemical reactions also form larger quantities of unwanted chemicals, like formaldehyde. Unfortunately, the ozone does not react to reduce other unwanted chemicals like nicotine, benzene, acrolein and many others.
Many manufacturers of ozone generators, ionizers and electronic precipitators suggest in their literature and their TV and radio commercials that the ozone created by their products attacks and destroys the bad things in indoor air and leaves the good. The reality is that when ozone is introduced into an indoor environment, the results are often not anticipated, predictable or beneficial.
Bibliography
Esswein, Eric J. and Boeniger.; “Effects of an ozone-generating air-purifying device on Reducing Concentrations Of Formalehyde in Air.” Applied Occupational Environmental Hygiene 9(2): 136-146, 1994
Mason, Mark, Xiaoyu Liu, Krebs, Kenneth; Sparks, Leslie; “Full-Scale Chamber Investigation of Air Freshener Emissions”; Journal of Environmental Science and Technology, 2004.
Morrison, G.C. and W.W. Nazaroff; “Ozone interactions with carpet: secondary emissions of aldehydes.” Environmental Science Technology 36: 2185-2192, 2002.
Sarwar, Golam, Olson, D. A., Corsi, Richard L., Weschler, Charles J.; “Indoor fine particles: The Role of Terpene Emissions from Consumer Products. Journal of Air and Waste Management 54: 367-377, 2004.
Weschler, C. J., A. T. Hodgson and J. D. Wooley; “Indoor Chemistry: Ozone, Volatile organic compounds and carpets.” Environmental Science Technology 26: 2371-2377, 1992.